Area Calculations
RECTANGLES
The area of a rectangle is the product of the base length and its height.
Example: A rectangle whose length is 8 metres has an area of 32 square metres. Calculate the width.
Solution:
Let the width of the rectangle be x
Formula: A = l × w
32 = 8 × x (dividing both sides by 8)
x = 4 metres
SQUARES
A square is a special rectangle where all sides are equal.
Formula: A = side × side
Example: Calculate the area of the square drawn below.
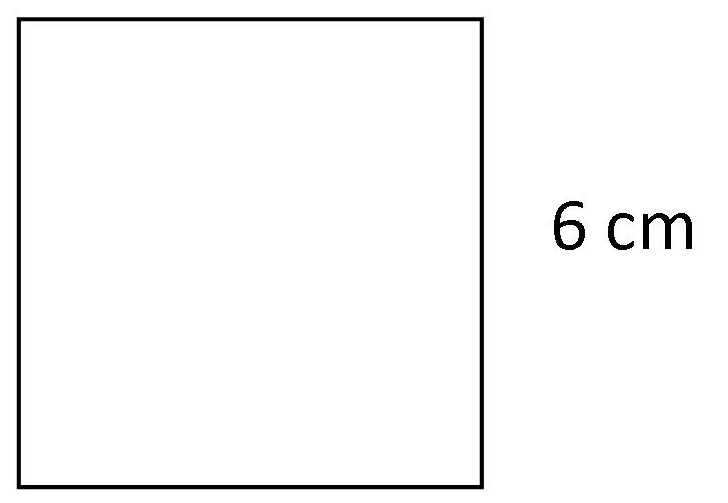
Solution:
Formula: A = s × s
A = 6 cm × 6 cm
Answer: A = 36 cm²
TRIANGLES
The area of a triangle is A = ½(base × height). The base and height always make an angle of 90°.
Example: Calculate the area of the triangle shown below.
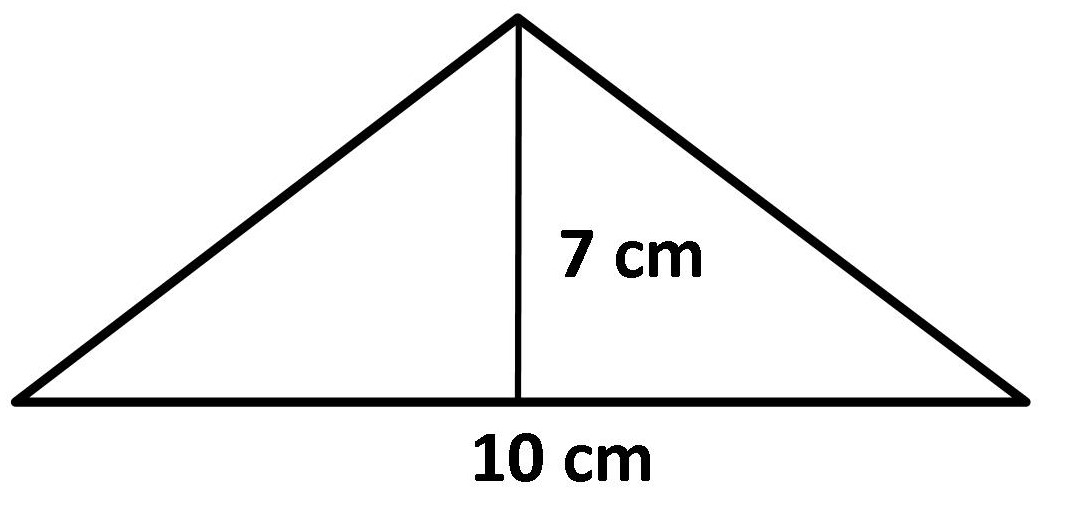
Solution:
Formula: A = ½(base × height)
A = ½ (10 × 7)
Answer: A = 35 cm²
TRAPEZIUM
A trapezium is a four-sided figure in which two sides are parallel. The area of a trapezium is given by the formula:
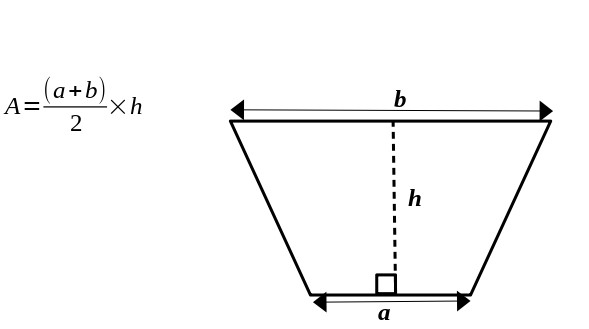
Example:
Find the area of the trapezium shown below.
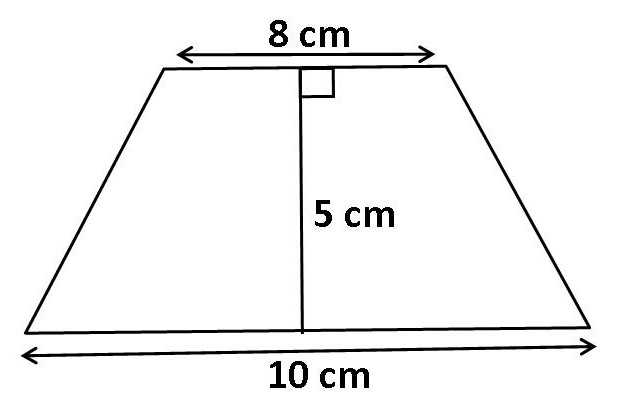
Solution:
The formula for the area of a trapezium is:
\[ A = \frac{(a + b)}{2} \times h \]
Where:
- a = 10 cm
- b = 8 cm
- h = 5 cm
Now, substituting the values:
\[ A = \frac{(10 + 8)}{2} \times 5 \]
\[ A = \frac{18}{2} \times 5 \]
\[ A = 9 \times 5 = 45 \, \text{cm}^2 \]
CIRCLES
The area of a circle is given by the formula:
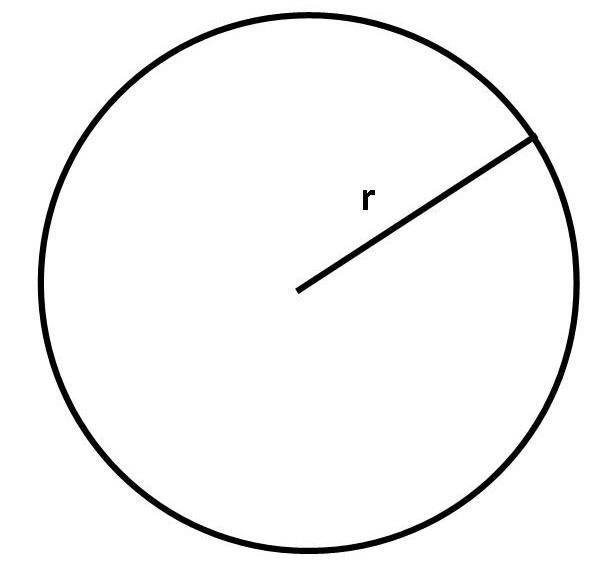
Formula: A = π × r² where r is the radius of the circle.
Example: A circle has a diameter of 70 m, calculate the area of the circle.
Solution: The diameter = 70 m, so the radius = 35 m.
Formula: A = π × r²
A = π × (35)²
Answer: A = 3,850 m²
AREA OF IRREGULAR OBJECTS
The steps are:
- Count Full Squares
- Count Partial Squares
- Find the area using the formula:
Formula: Area = ((Number of Full Squares) + (Partial Squares))/2
Example: Find the area of the figure below.
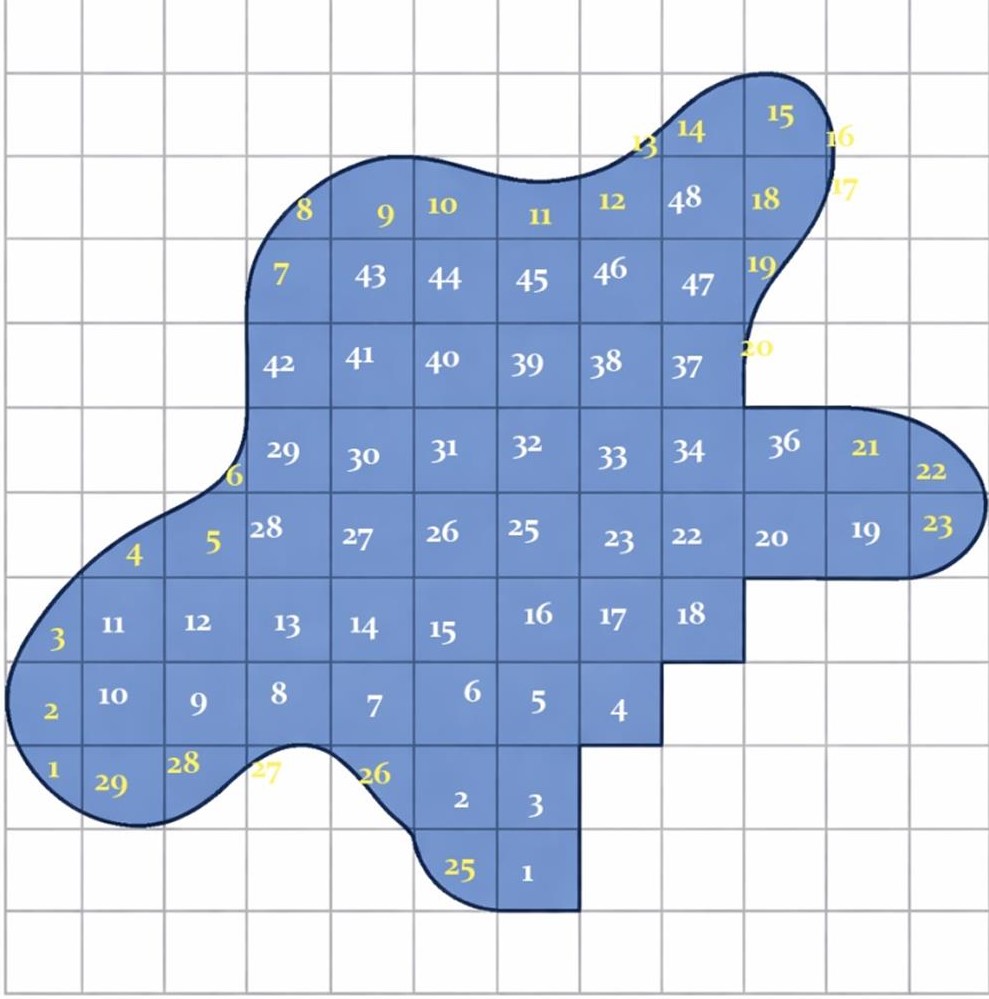
Solution:
The formula for calculating the area is:
\[ A = \text{no. of complete squares} + \frac{\text{no. of incomplete squares}}{2} \]
Where:
- Complete squares = 48
- Incomplete squares = 29
Now, substituting the values:
\[ A = 48 + \frac{29}{2} \]
\[ A = 48 + 14.5 \]
\[ A = 62.5 \, \text{sq units} \]
VOLUME OF REGULAR OBJECTS
The table below shows the formulae used in calculating volumes of cubes, cuboids, and cylinders.
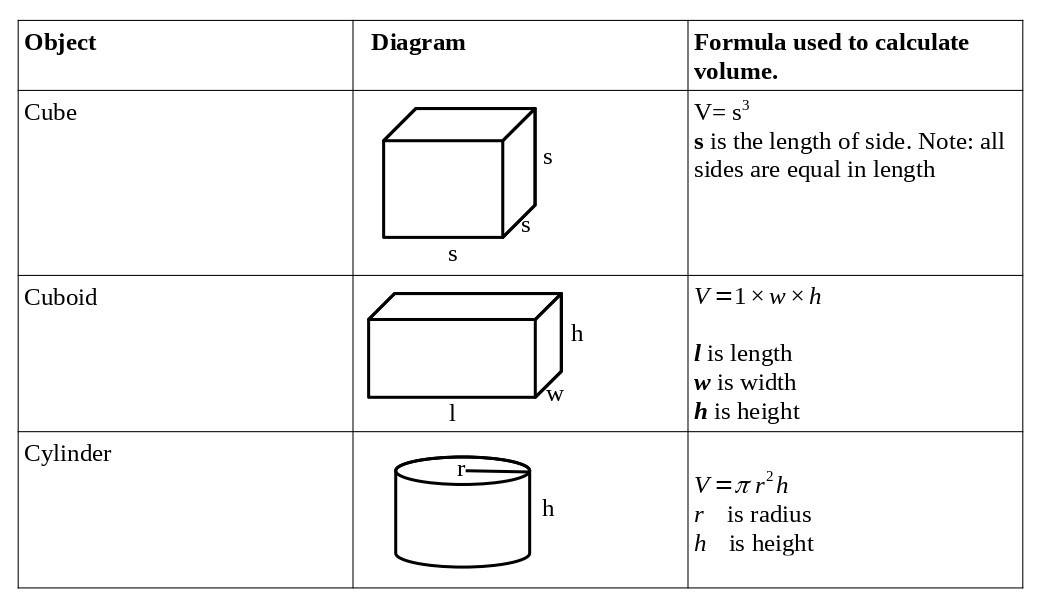
Example: Calculate the volume of a cube of side 5 cm.
Solution:
Volume of cube = s3
Volume = 5 cm × 5 cm × 5 cm = 125 cm3
Example: Calculate the volume of the cuboid measuring 140 cm by 30 cm by 70 cm in cubic meters.
Solution:
To get the answer in cubic meters, we convert all measurements to meters by dividing by 100:
Volume = (140/100) × (30/100) × (70/100) = 0.294 m3
Example: A cylindrical tank has a diameter of 140 cm and a height of 450 cm. Calculate the volume of the tank in cubic centimeters.
Solution:
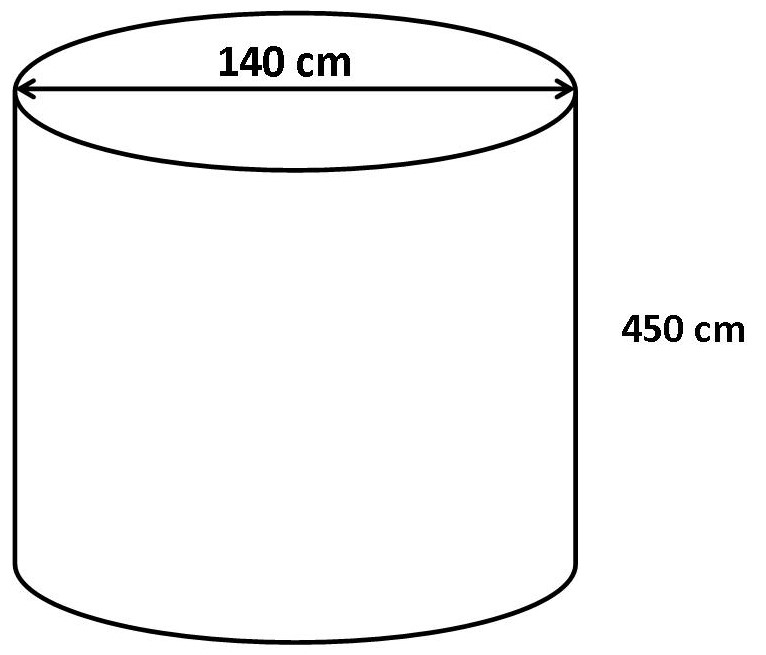
The diameter is 140 cm, therefore the radius is 70 cm.
Volume of cylinder = π × 702 × 450 = 6 930 000 cm3
Density
Definition
- Density is the mass of a substance per unit volume.
- It tells us how tightly matter is packed in a substance.
- It is a physical property of matter.
Formula:
The formula for density is:
Density (ρ) = Mass (m) / Volume (V)
SI Unit
The SI unit of density is kilogram per cubic meter (kg/m3).
Other common units include:
- grams per cubic centimeter (g/cm3)
- 1 g/cm3 = 1000 kg/m3
Rearranged Formulas
Sometimes, you can calculate mass or volume if density is known:
To calculate Mass, use:
Mass (m) = Density (ρ) × Volume (V)
To calculate Volume, use:
Volume (V) = Mass (m) / Density (ρ)
Example Calculations
Example 1: A substance has a mass of 200 g and a volume of 50 cm3. Find its density.
Solution:
Density = Mass / Volume = 200 g / 50 cm3 = 4.00 g/cm3
Example 2: A block of metal has a volume of 0.02 m3 and a density of 8000 kg/m3. Find its mass.
Solution:
Mass = Density × Volume = 8000 kg/m3 × 0.02 m3 = 160 kg
Determining Density of Irregular Objects Using Eureka Method
1. Principle
The Eureka method (also called the water displacement method) is based on Archimedes' principle: An object submerged in a fluid displaces a volume of fluid equal to its own volume. This method is used when the object does not have a regular shape, so its volume cannot be measured using a formula.
2. Apparatus
- Eureka can (overflow can)
- Measuring cylinder
- Thread or string
- Balance (to measure mass)
- Irregular object
3. Procedure
- Measure the mass of the object using a balance.
- Fill the Eureka can with water until it reaches the spout.
- Suspend the object using a thread and gently lower it into the Eureka can without touching the sides.
- Collect the displaced water in a measuring cylinder.
- Measure the volume of the displaced water. Volume of water displaced = volume of the object.
- Calculate the density using the formula: Density = Mass / Volume.
4. Example: An irregular rock has a mass of 300 g. When submerged in a Eureka can, it displaces 100 cm3 of water.
Solution:
Density = Mass / Volume = 300 g / 100 cm3 = 3.00 g/cm3
5. Key Points / Tips
- The object must be fully submerged.
- Avoid splashing to get an accurate volume measurement.
- Ensure the measuring cylinder measures displaced water accurately.
- This method works for solids that do not dissolve in water.
Speed
1. Definition
- Speed is the distance traveled by an object per unit time.
- It tells us how fast an object is moving, without considering direction.
- It is a scalar quantity (has magnitude only).
2. Formula:
The formula for speed is:
Speed (v) = Distance (d) / Time (t)
3. SI Unit
The SI unit of speed is meter per second (m/s).
Other common units include:
- Kilometers per hour (km/h) → 1 m/s = 3.6 km/h
- Centimeters per second (cm/s)
4. Rearranged Formulas
If speed, distance, or time is unknown, you can calculate it as:
To calculate Speed (v):
v = d / t
To calculate Distance (d):
d = v × t
To calculate Time (t):
t = d / v
5. Example Calculations
Example 1: A car travels 150 km in 3 hours. Find its speed.
Solution:
Speed = Distance / Time = 150 km / 3 hours = 50 km/h
Example 2: A cyclist travels at 5 m/s for 20 seconds. Find the distance covered.
Solution:
Distance = Speed × Time = 5 m/s × 20 s = 100 meters
6. Key Points
- Speed is a scalar (only magnitude, no direction).
- Make sure distance and time units are compatible before calculating.
Summary
Introduction
- Laboratory work involves the use of different apparatus and scientific skills.
- Accurate handling and observation improve experiment results.
- Science learning combines theory and practical activities.
Basic Skills in Science
- Manipulative skills involve handling apparatus correctly.
- Observation skills use senses to gather information.
- Measurement skills require correct units and tools.
- Classification groups items based on similarities.
- Prediction suggests possible outcomes.
- Communication records and shares results.
- Conclusion gives final answers based on findings.
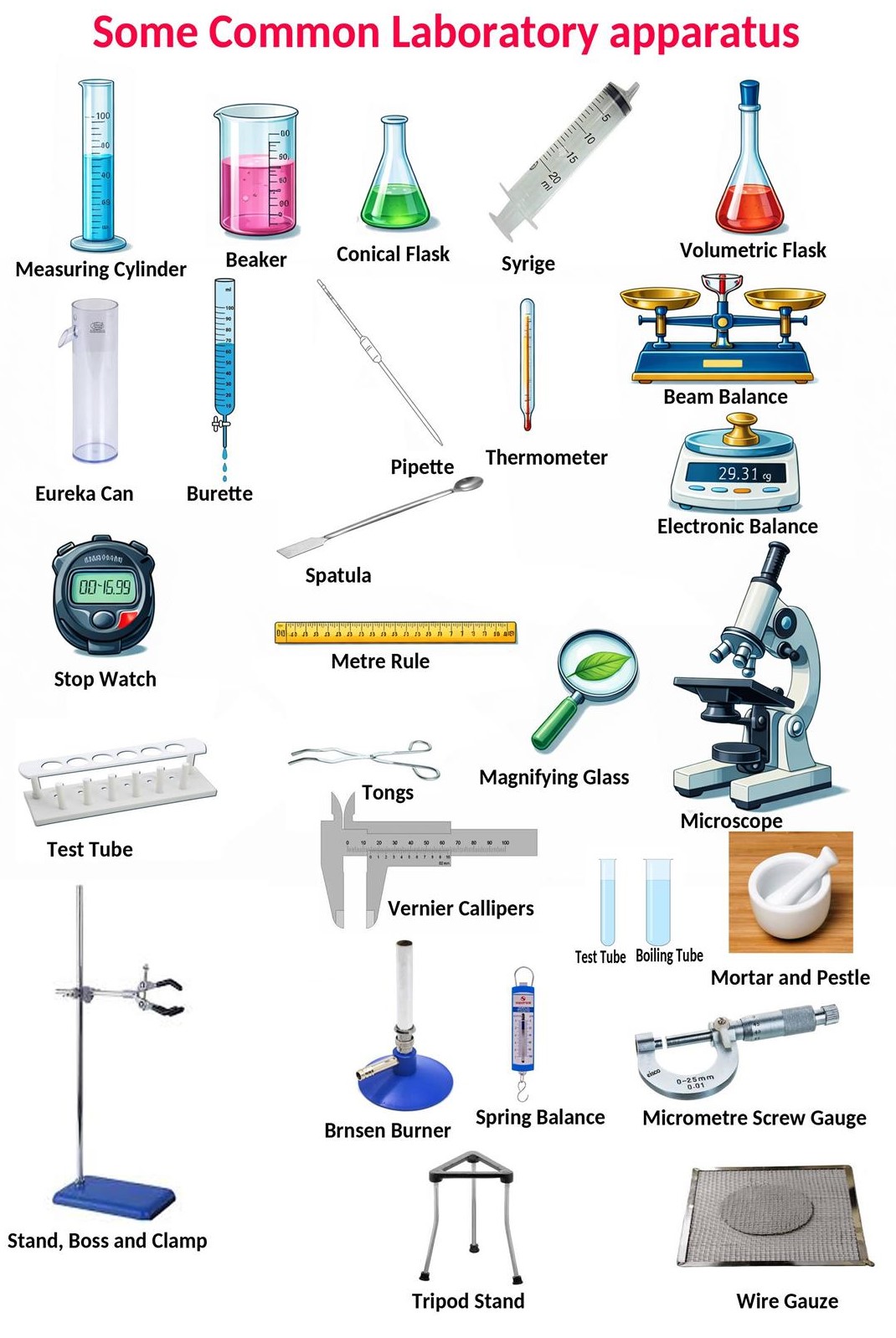
Laboratory Apparatus
- Different apparatus are used for measuring volume, mass, temperature, length, and time.
- Each apparatus has a specific function in experiments.
- Correct use ensures accurate and reliable results.
Care of Apparatus
- Handle apparatus carefully to avoid damage.
- Clean equipment after use.
- Store apparatus properly in safe, dry places.
- Proper care increases durability and accuracy.
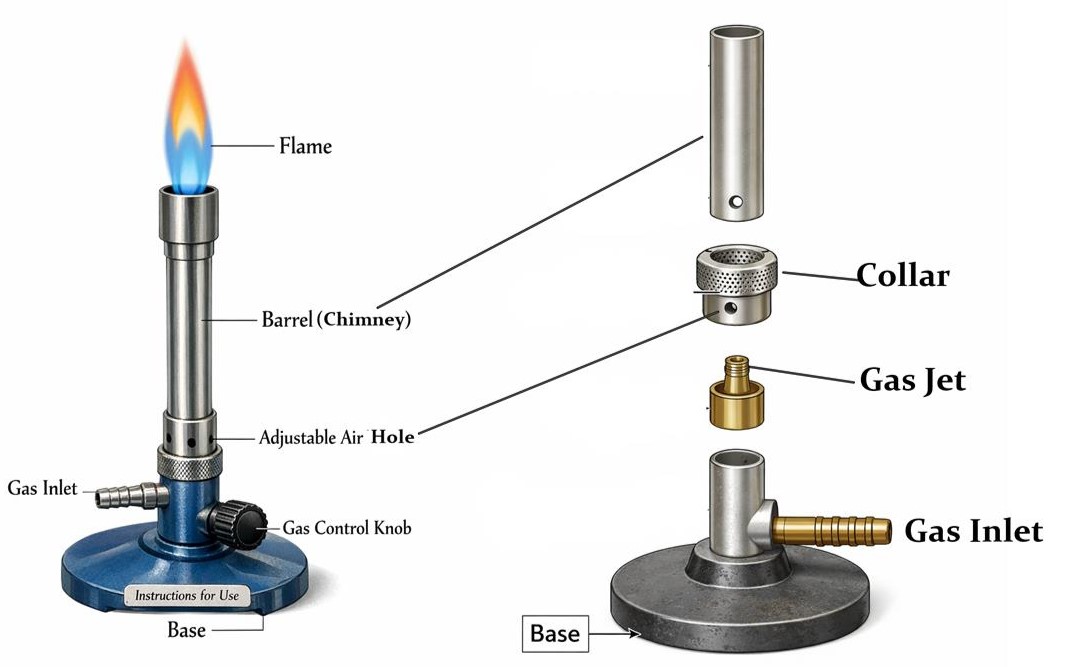
Bunsen Burner
- Used for heating substances in the laboratory.
- Has parts such as base, barrel, air hole, collar, and gas inlet.
- Produces yellow (safety) and blue (heating) flames.
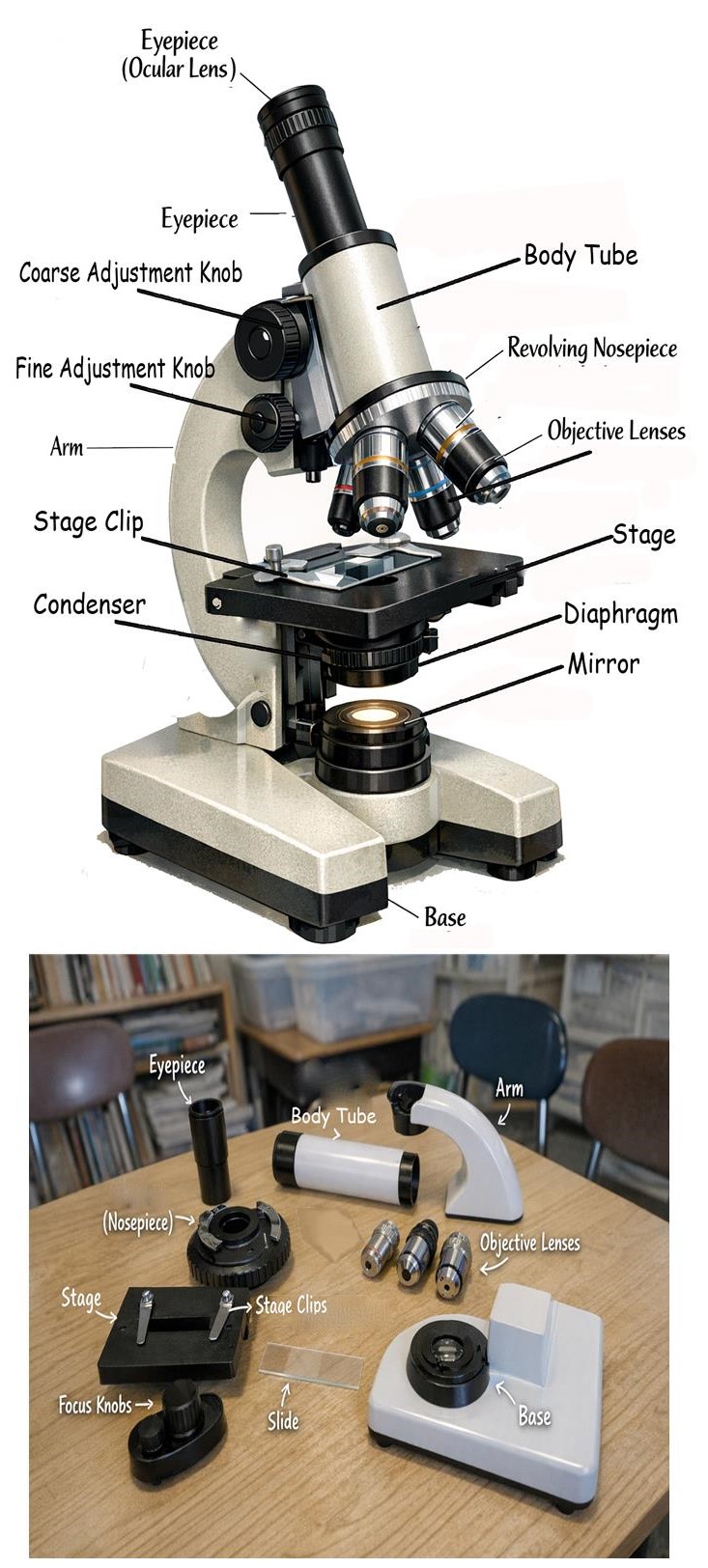
Light Microscope
- Used to magnify very small objects.
- Main parts include eyepiece, objective lenses, stage, and adjustment knobs.
- Proper handling and cleaning ensure clear observation.
SI Units
- SI units are standard units used worldwide in science.
- Examples include metre (m), kilogram (kg), and second (s).
- They ensure consistency and accuracy in measurements.
Derived Quantities
- Derived quantities are obtained from basic quantities.
- Examples include area, volume, speed, and density.
- They involve calculations using standard units.
Importance of SI Units
- Ensure uniformity in scientific measurements.
- Help in accurate calculations and comparisons.
- Used globally in science, engineering, and industry.
Practical Activities
- Experiments help apply scientific knowledge.
- Develop skills such as measuring, observing, and recording.
- Improve understanding through hands-on experience.
Assessment
Laboratory Science Assessment
Answer all 20 questions on laboratory apparatus, science skills, SI units, and measurements.
Certificate of Achievement
This certifies that
has successfully completed the assessment on
Laboratory Science
Score: